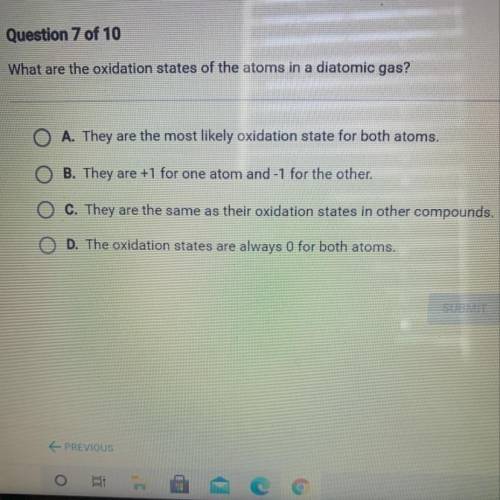
What are the oxidation states of the atoms in a diatomic gas?
...

Chemistry, 26.11.2020 20:40, myla18jabbar
What are the oxidation states of the atoms in a diatomic gas?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 12:00, kayla32213
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1

Chemistry, 22.06.2019 17:00, brownvester44
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Mathematics, 13.10.2020 15:01


Mathematics, 13.10.2020 15:01

Physics, 13.10.2020 15:01

Spanish, 13.10.2020 15:01

History, 13.10.2020 15:01



Business, 13.10.2020 15:01






