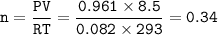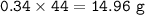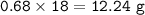Chemistry, 26.11.2020 03:00, kell22wolf
If 8.50 L of natural gas, which is essentially methane (CH4), undergoes complete combustion at 730 mm Hg and 20 degrees C, how many grams of each product are formed?
Grams of CO2=
Grams of H2O=

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, Zagorodniypolina5
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 23.06.2019 01:00, daniel1480
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2

Chemistry, 23.06.2019 02:00, Paytonsmommy09
Butane gas reacts with oxygen gas to give carbon dioxide gas and water vapor (gas). if you mix butane and oxygen in the correct stoichiometric ratio, and if the total pressure of the mixture is 390 mmhg, what is the pressure (in mmhg) of water vapor after the reaction is completed (temperature and volume do not change).
Answers: 2
Do you know the correct answer?
If 8.50 L of natural gas, which is essentially methane (CH4), undergoes complete combustion at 730 m...
Questions in other subjects:


History, 25.07.2021 23:00


English, 25.07.2021 23:00



Mathematics, 25.07.2021 23:00

English, 25.07.2021 23:00


Mathematics, 25.07.2021 23:00