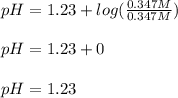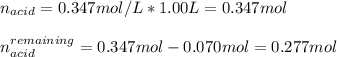Chemistry, 24.11.2020 21:00, carliehanson9908
A buffer solution is made that is 0.347 M in H2C2O4 and 0.347 M KHC2O4.
1. If Ka for H2C2O4 is 5.90E^-2, what is the pH of the buffer solution?
b. Write the net ionic equation for the reaction that occurs when 0.070 mol KOH is added to 1.00 L of the buffer solution.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, pinkycupcakes3oxbqhx
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 08:20, pilarmonsivais
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3

Chemistry, 22.06.2019 08:30, mosthatedpicky1
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 10:30, jahmira96
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2
Do you know the correct answer?
A buffer solution is made that is 0.347 M in H2C2O4 and 0.347 M KHC2O4.
1. If Ka for H2C2O4 is 5.90...
Questions in other subjects:




Mathematics, 08.01.2020 02:31




Chemistry, 08.01.2020 02:31


History, 08.01.2020 02:31



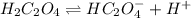
![pH=pKa+log(\frac{[base]}{[acid]} )](/tpl/images/0926/6955/33848.png)