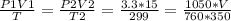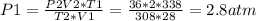2) A gas takes up a volume of 15 liters, has a pressure of 3.3 atm, and a temperature of
299 K. If I raise the temperature to 350 K and lower the pressure to 1050 mmHg, what is the
new volume of the gas?
3) A gas that has a volume of 28 liters, a temperature of 65 °C, and an unknown pressure
has its volume increased to 36 liters and its temperature decreased to 35 °C. If I measure the
pressure after the change to be 2.0 atm, what was the original pressure of the gas?
work too pls

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, eamccoy1
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2


Chemistry, 22.06.2019 09:00, kkmonsterhigh18
The diagram below shows a cell placed in a solution. a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution. only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it. it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3
Do you know the correct answer?
2) A gas takes up a volume of 15 liters, has a pressure of 3.3 atm, and a temperature of
299 K. If...
Questions in other subjects:

Mathematics, 17.09.2020 09:01

Chemistry, 17.09.2020 09:01

Mathematics, 17.09.2020 09:01

Mathematics, 17.09.2020 09:01

Mathematics, 17.09.2020 09:01

Mathematics, 17.09.2020 09:01

Mathematics, 17.09.2020 09:01

Mathematics, 17.09.2020 09:01

Physics, 17.09.2020 09:01

Mathematics, 17.09.2020 09:01