
Chemistry, 22.11.2020 21:30, LtotheJ0225
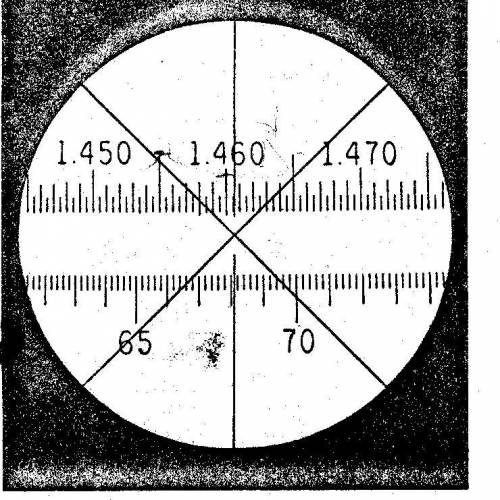
How do you determine the scale number on the refractive index? What makes this 1.4606? How did they get that 6 at the end? Like how are the last two numbers determined if the 06 weren’t there if it was just 1.46?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 15:00, NatalieKnows
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1
Do you know the correct answer?
How do you determine the scale number on the refractive index? What makes this 1.4606? How did they...
Questions in other subjects:

Mathematics, 18.02.2021 16:40





Mathematics, 18.02.2021 16:40



Mathematics, 18.02.2021 16:40






