
Chemistry, 21.11.2020 04:00, dallisryan
2.911 grams of ethanol C2H5OH is burned and produces how many grams of water vapor? Please help me or I'm dead.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 00:20, destromero
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1

Chemistry, 23.06.2019 01:30, koggebless
The solubility of barium nitrate is 9.02 g/100 g h2o at 20°c. a 15.2 g sample of barium nitrate is added to 200.0 g of water at 20°c. is the solution saturated, unsaturated, or supersaturated? a. unsaturated b. saturated c. supersaturated
Answers: 1
Do you know the correct answer?
2.911 grams of ethanol C2H5OH is burned and produces how many grams of water vapor?
Please help me...
Questions in other subjects:


Biology, 06.07.2019 20:30

English, 06.07.2019 20:30

Physics, 06.07.2019 20:30


English, 06.07.2019 20:30


Mathematics, 06.07.2019 20:30



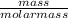
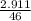 = 0.06mole
= 0.06mole 



