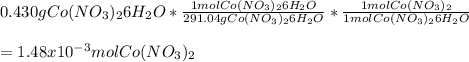Chemistry, 20.11.2020 14:00, meramera50
If you mass 0.430 g of Co(NO3)2 6H2O and dissolve it in water, what further information would you need to calculate:.
a. the number of moles of Co(NO3)2 in the solution?
b. the molarity of the solution

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, jivsf
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3


Chemistry, 23.06.2019 07:30, mazielynn84
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes or no?
Answers: 1

Chemistry, 23.06.2019 09:00, hunterwilliams375
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate according to the following equation:
Answers: 2
Do you know the correct answer?
If you mass 0.430 g of Co(NO3)2 6H2O and dissolve it in water, what further information would you ne...
Questions in other subjects:



Mathematics, 11.12.2020 06:50


Mathematics, 11.12.2020 06:50

Mathematics, 11.12.2020 06:50


Mathematics, 11.12.2020 06:50

Mathematics, 11.12.2020 06:50