Look at the balanced equation below:
C2H6O + 3 O2 → 2 CO2 + 3 H2O
If you produced 11.5...


Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:00, hannah5143
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

Chemistry, 22.06.2019 13:10, dookiefadep5n1tt
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1


Do you know the correct answer?
Questions in other subjects:

SAT, 24.03.2020 21:05


Physics, 24.03.2020 21:05



Mathematics, 24.03.2020 21:05




Biology, 24.03.2020 21:05


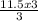 = 11.5moles of oxygen gas
= 11.5moles of oxygen gas



