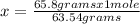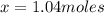B 0 10.9 mol Cu

Chemistry, 18.11.2020 23:50, deedee2616
How many moles of copper (Cu) are in 65.8 g Cu? (3 points)
A 01.04 mol Cu
B 0 10.9 mol Cu
C 41.7 mol Cu
D 63.5 mol Cu

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 22:40, lindseyklewis1p56uvi
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization. a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution. part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 00:00, scottykinkade7860
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1

Chemistry, 23.06.2019 05:00, jjoyner
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2
Do you know the correct answer?
How many moles of copper (Cu) are in 65.8 g Cu? (3 points)
A 01.04 mol Cu
B 0 10.9 mol Cu
B 0 10.9 mol Cu
Questions in other subjects:



English, 20.05.2021 18:20


French, 20.05.2021 18:20


Mathematics, 20.05.2021 18:20

History, 20.05.2021 18:20

Biology, 20.05.2021 18:20


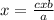
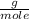 , this is the amount of mass a substance contains in one mole.
, this is the amount of mass a substance contains in one mole.