Please Help! I am in desperate need
Question 1
Answer the true/false questions based on the d...

Chemistry, 18.11.2020 23:20, dthompson365
Please Help! I am in desperate need
Question 1
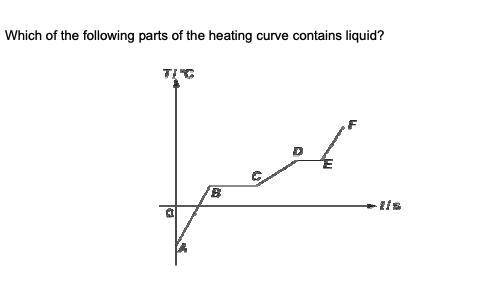
Answer the true/false questions based on the diagram above.
I have no clue where the graph is tho, my teacher did not post one.
Addition of a catalyst will change the value for ΔH
True
False
Question 2
Consider the chemical reaction equation shown.
H + H--> H-H
Is this reaction endothermic or exothermic?
a
Endothermic
b
Exothermic
Question 3
a
AB
b
BC
c
CD
d
DE
Question 4
A chemical reaction has a reaction enthalpy of −512 kJ/mol. Is it endothermic or exothermic?
a
Endothermic
b
Exothermic
Question 5
What is the energy required to evaporate two moles of liquid water given the following equations?
2H2(g) + O2(g) → 2H2O(g) ∆H = -483.6 kJ
2H2(g) + O2(g) → 2H2O(l) ∆H = -571.6 kJ
a
44.0 kJ
b
88.0 kJ
c
527.6 kJ
d
1055.2 kJ

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:50, britotellerialuis
Evaluate this exponential expression,8. (2 + 3)2 – 42
Answers: 3

Chemistry, 22.06.2019 01:20, sarinaneedshelp01
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1

Chemistry, 22.06.2019 20:10, sarahalexa19
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Mathematics, 06.10.2019 10:30















