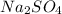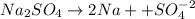Chemistry, 18.11.2020 16:50, kaytlynnmeyer
If a 3.0 M solution of glucose (C6H12O6), a 2.0 M solution of Na2SO4 and a 1.0 M solution of (NH4)3PO4, is made, which solution will have the lowest vapor pressure, highest boiling point, and lowest freezing point?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:00, eweqwoewoji
Agood hypothesis includes which of the following? a: prediction b: data c: uncertainty d: conclusion
Answers: 1

Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Do you know the correct answer?
If a 3.0 M solution of glucose (C6H12O6), a 2.0 M solution of Na2SO4 and a 1.0 M solution of (NH4)3P...
Questions in other subjects:





Chemistry, 20.10.2021 19:10

Chemistry, 20.10.2021 19:10




English, 20.10.2021 19:10