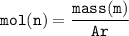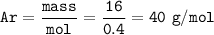Chemistry, 18.11.2020 14:00, joycewingate919
calculate the relative atomic mass of calcium given that the number of moles in 0.4 moles and mass is 16 g

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, penny3109
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 21.06.2019 18:10, hunterthompson2
Which is true of transition metals when moving from left to right on the periodic table? the d sublevels are not filled across the period. the cation radii become larger across the period. atomic radii increase slightly and then start to decrease. atomic radii decrease slightly and then start to increase. o
Answers: 2


Chemistry, 22.06.2019 09:40, keiracoles
How many grams of aluminum will there be in 98g of al2o3?
Answers: 1
Do you know the correct answer?
calculate the relative atomic mass of calcium given that the number of moles in 0.4 moles and mass i...
Questions in other subjects:

Mathematics, 11.01.2021 20:40

Health, 11.01.2021 20:40

Mathematics, 11.01.2021 20:40

Mathematics, 11.01.2021 20:40

English, 11.01.2021 20:40

English, 11.01.2021 20:40

English, 11.01.2021 20:40


Mathematics, 11.01.2021 20:40

Mathematics, 11.01.2021 20:40