
Chemistry, 18.11.2020 06:50, anyone2562
Calculate the chemical amount of ammonia present in a 11.22g sample of the compound

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 23:50, josie311251
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 01:00, carson9373
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1

Chemistry, 23.06.2019 08:00, mackaylabarnes22
Ineed this awnser fast select the correct answer. this chemical equation represents the burning of methane, but the equation is incomplete. what is the missing coefficient in both the reactants and the products? ch4 + → co2 + a. 0 b. 1c. 2d. 3 e. 4
Answers: 3
Do you know the correct answer?
Calculate the chemical amount of ammonia present in a 11.22g sample of the compound...
Questions in other subjects:

Mathematics, 20.06.2021 17:20

Mathematics, 20.06.2021 17:20

History, 20.06.2021 17:20


Mathematics, 20.06.2021 17:20


English, 20.06.2021 17:20


Mathematics, 20.06.2021 17:20

Geography, 20.06.2021 17:20


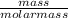
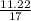 = 0.66mole
= 0.66mole 



