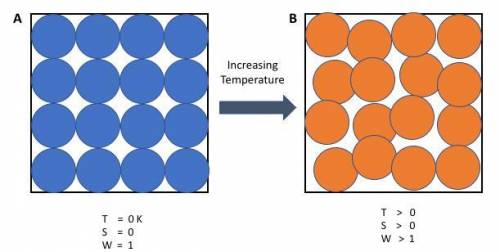
Chemistry, 17.11.2020 17:40, Hilljos018
Calculate ΔS∘rxn for the following reaction. The ΔS∘ for each species is shown below the reaction.
C2H2(g) + 2H2(g) → C2H6(g)
S∘(J/mol⋅K) 200.9 130.7 229.2
A) -159.0 J/K
B) -233.1 J/K
C) +233.1 J/K
D) 303.3 J/K
E) 560.8 J/K

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, larreanathalie3523
The diagram shows the structures of horse and cat forelimbs. what does the diagram suggest about the evolutionary relationship between these two mammals? a. they have homologous structures, indicating a common ancestor. b. they have analogous structures, indicating a common ancestor. c. they have homologous structures, indicating that they do not have a common ancestor. d. they have analogous structures, indicating that they do not have a common ancestor.
Answers: 2


Chemistry, 22.06.2019 21:30, MJyoungboy
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 23.06.2019 02:20, theactualslash
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2
Do you know the correct answer?
Calculate ΔS∘rxn for the following reaction. The ΔS∘ for each species is shown below the reaction....
Questions in other subjects:

Mathematics, 25.08.2021 14:00

Social Studies, 25.08.2021 14:00

Mathematics, 25.08.2021 14:00


Mathematics, 25.08.2021 14:00




Business, 25.08.2021 14:00