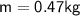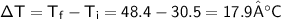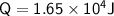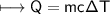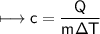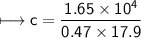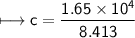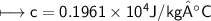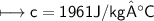Chemistry, 13.11.2020 09:10, naeaamm2528
A chemist carefully measures the amount of heat needed to raise the temperature of a 0.47 kg sample of C6H7N from 30.5 degrees C to 48.4 degrees C. The experiment shows that 1.65 x 10^4 J of heat are needed. What can the chemist report for the molar heat capacity of C6H7N? Be sure your answer has the correct number of significant digits.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, giiffnlojd
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 04:00, breannaasmith1122
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1
Do you know the correct answer?
A chemist carefully measures the amount of heat needed to raise the temperature of a 0.47 kg sample...
Questions in other subjects:

Mathematics, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01



Mathematics, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01


Mathematics, 20.10.2020 09:01


Business, 20.10.2020 09:01