
Chemistry, 12.11.2020 07:30, villatoroo84502
If a reaction generated 3.5 g of H2O how many moles of water were formed?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:50, toniawu18
Problem page gaseous ethane reacts with gaseous oxygen gas to produce gaseous carbon dioxide and gaseous water . if of water is produced from the reaction of of ethane and of oxygen gas, calculate the percent yield of water. be sure your answer has the correct number of significant digits in it.
Answers: 2

Chemistry, 21.06.2019 22:30, officialgraciela67
Embryos of different species look very similar, which shows that the organisms share a ancestor.
Answers: 1

Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
Do you know the correct answer?
If a reaction generated 3.5 g of H2O how many moles of water were formed?...
Questions in other subjects:

Mathematics, 09.11.2020 19:50





Mathematics, 09.11.2020 19:50



Mathematics, 09.11.2020 19:50


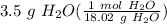 = 0.194229 mol H₂O
= 0.194229 mol H₂O




