__Ag2S --> __Ag + __S8


Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:50, JuniperGalaxy
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 04:00, speris1443
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 09:50, bridgetosanders
What are four significant sources of ghgs that come from wostem washington?
Answers: 2
Do you know the correct answer?
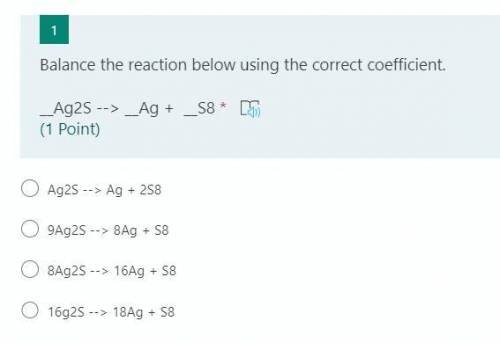
Balance the reaction below using the correct coefficient.
__Ag2S --> __Ag + __S8
__Ag2S --> __Ag + __S8
Questions in other subjects:


Computers and Technology, 17.11.2020 18:00

Mathematics, 17.11.2020 18:00

Mathematics, 17.11.2020 18:00

Mathematics, 17.11.2020 18:00


Physics, 17.11.2020 18:00

Mathematics, 17.11.2020 18:00


Mathematics, 17.11.2020 18:00