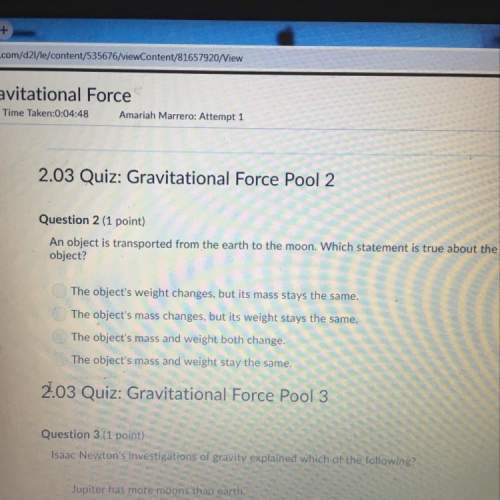
How does the law of conservation of mass apply to this reaction: CH + O2 + 2H2O + 2002?
o Only the oxygen needs to be balanced. There are equal numbers of hydrogen and carbon
The law of conservation of mass has already been applied. There is an equal number of each element on both sides of
O the equation
Each element needs to be balanced
O
o
The equation needs to be balanced. There are fewer oxygen atoms in the equation than hydrogen or carbon

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 05:50, starfox5454
Which of the following isotopes has the same number of neutrons as phosphorus-31?
Answers: 1

Chemistry, 23.06.2019 13:30, gavianacandelar8522
The process by which liquid water changes into water vapour is called what ?
Answers: 2
Do you know the correct answer?
How does the law of conservation of mass apply to this reaction: CH + O2 + 2H2O + 2002?
o Only the...
Questions in other subjects:



Mathematics, 23.01.2021 01:30


English, 23.01.2021 01:30

Mathematics, 23.01.2021 01:30



Advanced Placement (AP), 23.01.2021 01:30

Mathematics, 23.01.2021 01:30