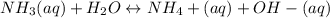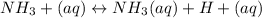A) Earlier you were told that of all of the 14 solutes you will be studying, the only one that is not appreciably ionized in water is aqueous ammonia. What does this statement imply about the equilibrium point of the reaction involving aqueous ammonia/ammonium hydroxide?B) Use your results and conclusions from part (a) to explain why a complicated name like Aqueous ammonia/ammonium hydroxide is used for this solution?

Answers: 2
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2
Do you know the correct answer?
A) Earlier you were told that of all of the 14 solutes you will be studying, the only one that is no...
Questions in other subjects:

Mathematics, 30.08.2021 03:10



Mathematics, 30.08.2021 03:20

Mathematics, 30.08.2021 03:20

History, 30.08.2021 03:20