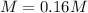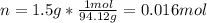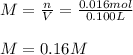Chemistry, 11.11.2020 17:50, jessica6597
Phenol (C6H5OH) is often used as an antiseptic in mouthwashes and throat lozenges. If a mouthwash has a phenol concentration of 1.5 g per 100 mL of solution, what is the molarity of phenol?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:20, merrickrittany
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 18:50, christhegreat1
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1


Chemistry, 22.06.2019 23:30, johnnysteeler9934
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
Do you know the correct answer?
Phenol (C6H5OH) is often used as an antiseptic in mouthwashes and throat lozenges. If a mouthwash ha...
Questions in other subjects:

Mathematics, 25.06.2019 00:00

English, 25.06.2019 00:00

History, 25.06.2019 00:00

History, 25.06.2019 00:00



Mathematics, 25.06.2019 00:00

English, 25.06.2019 00:00