
Chemistry, 06.11.2020 17:10, delanieloya
Suppose that 0.410 mol of methane, CH4(g), is reacted with 0.560 mol of fluorine, F2(g), forming CF4(g) and HF(g) as sole products. Assuming that the reaction occurs at constant pressure, how much heat is released

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, 767sebmont
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2


Chemistry, 22.06.2019 12:00, vannitling12p4w44f
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 22.06.2019 18:50, cj31150631
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1
Do you know the correct answer?
Suppose that 0.410 mol of methane, CH4(g), is reacted with 0.560 mol of fluorine, F2(g), forming CF4...
Questions in other subjects:

Mathematics, 14.12.2021 17:10



Mathematics, 14.12.2021 17:10

Mathematics, 14.12.2021 17:10


Mathematics, 14.12.2021 17:10

Mathematics, 14.12.2021 17:10

Mathematics, 14.12.2021 17:10

Mathematics, 14.12.2021 17:10


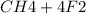 →
→ 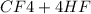
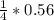 = 0.14mole of CF44 moles of fluorine → 4 moles of HF0.56 mole of fluorine → 0.56 mole of HF
= 0.14mole of CF44 moles of fluorine → 4 moles of HF0.56 mole of fluorine → 0.56 mole of HF



