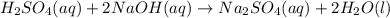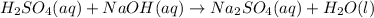Chemistry, 05.11.2020 19:10, slonekaitlyn01
Sulfuric acid, H2SO4, can be neutralized by sodium hydroxide, NaOH. The unbalanced equation is:
H2SO4(aq) + NaOH(aq) → Na2SO4(aq) + H2O(l)
A student who was asked to balance the reaction wrote the following:
H2SO4(aq) + Na2OH(aq) →Na2SO4(aq) + H3O(l)
Is this correct? Explain why or why not using what you know about the law of conservation of mass and chemical changes. If necessary, provide the correct balanced equation.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:50, kelonmazon2492
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3


Chemistry, 22.06.2019 17:40, Snowball080717
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 23.06.2019 04:00, Ezekielcassese
Which method would be best to separate a mixture of sand and gravel
Answers: 1
Do you know the correct answer?
Sulfuric acid, H2SO4, can be neutralized by sodium hydroxide, NaOH. The unbalanced equation is:
H2...
Questions in other subjects:

Mathematics, 28.08.2020 21:01


Mathematics, 28.08.2020 21:01

Mathematics, 28.08.2020 21:01

Mathematics, 28.08.2020 21:01



Mathematics, 28.08.2020 21:01

History, 28.08.2020 21:01