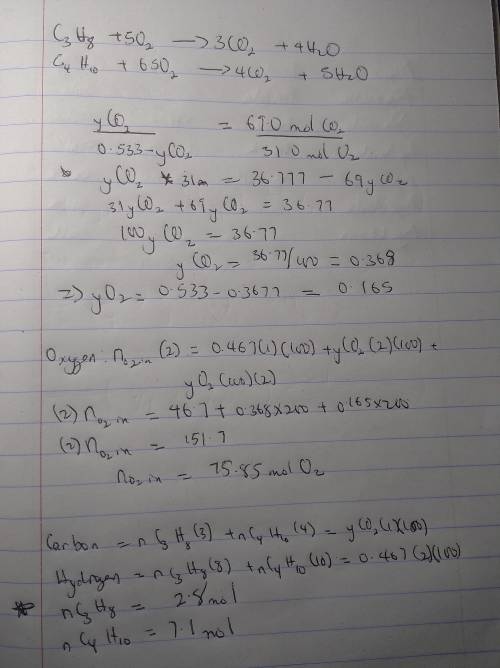Chemistry, 04.11.2020 19:00, tommyaberman
A mixture of propane and butane is burned with pure oxygen. The combustion products contain 46.7 mole% H2O. After all the water is removed from the products, the residual gas contains 69.0 mole% CO2 and the balance O2 a. What is the mole percent of propane in the fuel?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, Pizzapegasus1
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 07:10, angellong94
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 09:40, loveoneonly9153
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1
Do you know the correct answer?
A mixture of propane and butane is burned with pure oxygen. The combustion products contain 46.7 mol...
Questions in other subjects:

Mathematics, 21.06.2021 06:00


Business, 21.06.2021 06:00

Mathematics, 21.06.2021 06:00

English, 21.06.2021 06:00

Mathematics, 21.06.2021 06:00

Mathematics, 21.06.2021 06:00