Chemistry, 03.11.2020 21:00, cpcoolestkid4
Explain the relationship between the rate of effusion of a gas and its molar mass. Methane gas (CH4) effuses 3.4 times faster than an unknown gas. Determine the molar mass of the unknown gas. Show your work or explain your answer, giving specific values used to determine the answer.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:40, tatemelliott
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3


Chemistry, 22.06.2019 13:30, bryce99
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

Chemistry, 22.06.2019 13:30, hdhtvthjr
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1
Do you know the correct answer?
Explain the relationship between the rate of effusion of a gas and its molar mass.
Methane gas (CH4...
Questions in other subjects:


Chemistry, 01.07.2020 15:01


Physics, 01.07.2020 15:01


Mathematics, 01.07.2020 15:01






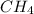 = 16 g/mole
= 16 g/mole