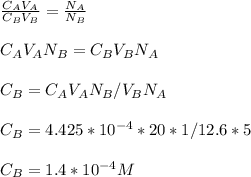Chemistry, 03.11.2020 03:20, sihamabdalla591
3.47 g of the hydrated "double salt", ammonium iron (II) sulfate hexahydrate, FeSO4(NH4)2SO4*6H2O was dissolved in 200. mL of water. 20.0 mL of the solution had some acid added to it and then it reacted completely with 12.6 mL of KMnO4 solution. Calculate the concentration of the KMnO4 solution given the full REDOX equation below. 5Fe2+ + MnO4- + 8H+ --> 5Fe3+ +Mn2+ + 4H2O

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 01:20, whrjegt4jrnfdvj
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 04:00, dustinsampsin2486
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3
Do you know the correct answer?
3.47 g of the hydrated "double salt", ammonium iron (II) sulfate hexahydrate, FeSO4(NH4)2SO4*6H2O wa...
Questions in other subjects:

Social Studies, 09.02.2020 20:18



Biology, 09.02.2020 20:18

History, 09.02.2020 20:18

World Languages, 09.02.2020 20:18

Mathematics, 09.02.2020 20:18


Mathematics, 09.02.2020 20:19


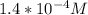
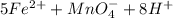 →
→ 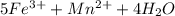
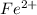 in FeSO₄(NH₄)₂SO₄*6H₂O
in FeSO₄(NH₄)₂SO₄*6H₂O 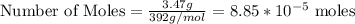
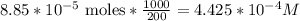
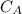 be concentration of
be concentration of 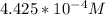
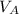 )= 20.0 ml
)= 20.0 ml
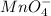 be
be 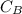 (the unknown)
(the unknown)
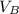 ) = 12.6 ml
) = 12.6 ml
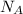 = 5 moles
= 5 moles
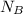 = 1 mole
= 1 mole