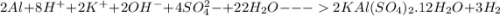Calculate the theoretical yield of potassium alum from 1.0000 g of aluminum, assuming that the aluminum is the limiting reagent. First, write the net reaction by adding the net ionic equations that represent the four steps in the synthesis:
(view image for reactions)
Write the net reaction by adding these four reactions and canceling out species that appear on both sides.


Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, Arealbot
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 20:30, Schoolworkspace453
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

Chemistry, 22.06.2019 22:00, jlegrand9098
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2
Do you know the correct answer?
Calculate the theoretical yield of potassium alum from 1.0000 g of aluminum, assuming that the alumi...
Questions in other subjects:

Computers and Technology, 27.10.2019 09:43

Mathematics, 27.10.2019 09:43

Mathematics, 27.10.2019 09:43

Mathematics, 27.10.2019 09:43

Biology, 27.10.2019 09:43

Arts, 27.10.2019 09:43



Arts, 27.10.2019 09:43