Chemistry, 29.10.2020 17:10, alexam2007
When 11.0g of Mg(s) reacts with excess water to produce 5.75g of Mg(OH)2, what is the percent yield of the reaction

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, Thomas7785
Two things that biomedical has invented or innovated
Answers: 1

Chemistry, 22.06.2019 11:00, snowprincess99447
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 12:30, quantamagic
Word equation for k(s)+h2o(l) yield koh(aq) + h2(g)
Answers: 1

Chemistry, 23.06.2019 04:31, diamondscott9297
Which of the following is an example of how telecommunication devices people do their jobs? a.) a security guard checks the time using a digital watch. b.) a banker does some quick math using a solar calculator. c.) a nurse uses a digital thermometer to take a patient’s temperature. d.) a construction worker reports in to his office using a cell phone.
Answers: 1
Do you know the correct answer?
When 11.0g of Mg(s) reacts with excess water to produce 5.75g of Mg(OH)2, what is the percent yield...
Questions in other subjects:

Geography, 02.08.2019 06:50





History, 02.08.2019 06:50


Business, 02.08.2019 06:50

Social Studies, 02.08.2019 06:50

Mathematics, 02.08.2019 06:50


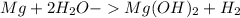
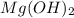
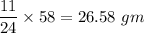 .
.