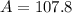Copying is not allowed. You must show all work in order to receive credit. Naturally occurring silver has two isotopes. Isotope A has a relative mass of 106.9051 and an abundance of 51.82%. Isotope B has a relative mass of 108.9047. Calculate the atomic mass of silver from these data.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:00, alaina3792
Of the groups of elements below, which are most likely to gain electrons to become anions? a. alkali metal b. boron group c. halogen d. transition metal
Answers: 2



Chemistry, 22.06.2019 22:30, medinajocelyn45
Which compound most likely has the greatest bond energy?
Answers: 2
Do you know the correct answer?
Copying is not allowed. You must show all work in order to receive credit. Naturally occurring silve...
Questions in other subjects:





History, 12.03.2020 20:19

Mathematics, 12.03.2020 20:19


History, 12.03.2020 20:19

Physics, 12.03.2020 20:19

Computers and Technology, 12.03.2020 20:19


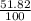
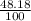
![A=\sum[(106.9051)\times \frac{51.82}{100})+(108.9047)\times \frac{48.18}{100}]]](/tpl/images/0851/2823/92a78.png)