
Chemistry, 26.10.2020 16:40, jsmith4184
What molar ratio of acetic acid to sodium acetate is required to create a buffer solution having a pH of 4.93 at 25°C? Ka for HC2H3O2 is 1.8 × 10–5.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, jescanarias22
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1


Chemistry, 22.06.2019 05:00, Angelanova69134
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Do you know the correct answer?
What molar ratio of acetic acid to sodium acetate is required to create a buffer solution having a p...
Questions in other subjects:


Business, 11.09.2019 19:30

Health, 11.09.2019 19:30




Biology, 11.09.2019 19:30



Mathematics, 11.09.2019 19:30


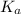 = Acid ionization constant of acetic acid =
= Acid ionization constant of acetic acid = 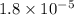
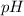 = Potential of hydrogen of buffer solution =
= Potential of hydrogen of buffer solution = 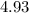
![K_a=\dfrac{[H+][A-]}{[HA]}\\\Rightarrow K_a=[H+]\dfrac{[A-]}{[HA]}](/tpl/images/0840/1638/61c39.png)
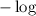 on both sides of the equation we get
on both sides of the equation we get![-\log K_a=-\log [H+]-\log\dfrac{[A-]}{[HA]}\\\Rightarrow pK_a=pH-\log\dfrac{[A-]}{[HA]}\\\Rightarrow pH=pK_a+\log\dfrac{[A-]}{[HA]}](/tpl/images/0840/1638/8d914.png)
![4.93=-\log(1.8\times 10^{-5})+\log\dfrac{[A-]}{[HA]}\\\Rightarrow 4.93-4.74=\log\dfrac{[A-]}{[HA]}\\\Rightarrow 0.19=\log\dfrac{[A-]}{[HA]}\\\Rightarrow 1.55=\dfrac{[A-]}{[HA]}\\\Rightarrow \dfrac{[HA]}{[A-]}=0.65](/tpl/images/0840/1638/a0e07.png)




