
Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1
Do you know the correct answer?
What is the net ionic equation of CuBr2+Na2S=CuS+2NaBr?...
Questions in other subjects:







Mathematics, 23.09.2020 05:01




 .
. . (Balanced.)
. (Balanced.) ,
, 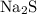 , and
, and  are all soluble in water.
are all soluble in water. is not soluble in water.
is not soluble in water. .
. .
. will be rewritten as
will be rewritten as  .
. .
.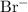 ions, andTwo
ions, andTwo 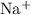 ions.
ions.




