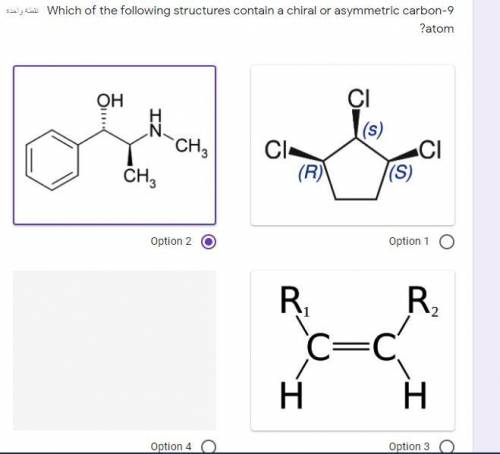
HELP PRETTY PLEASE
this question is driving me nuts itd be awesome if someone helped
...


Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 23.06.2019 01:30, jarteria0
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
Do you know the correct answer?
Questions in other subjects:





Mathematics, 10.04.2021 18:50

Mathematics, 10.04.2021 18:50

Mathematics, 10.04.2021 18:50

History, 10.04.2021 18:50

History, 10.04.2021 18:50

Social Studies, 10.04.2021 18:50