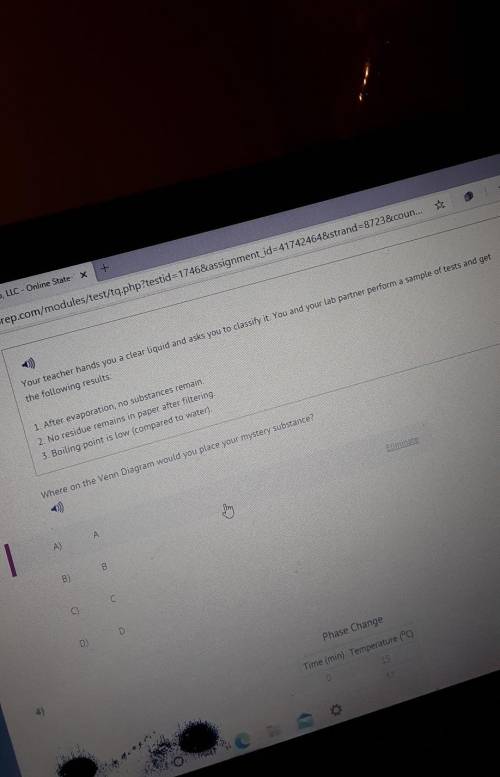
Where on the Venn Diagram would you place your mystery substance?
...

Chemistry, 22.10.2020 03:01, bibigardeax
Where on the Venn Diagram would you place your mystery substance?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, kristieroth1
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 04:00, heavyhearttim
4. absorption has the highest risk of overdose due to increased potency. a. rectal b. oral c. transdermal d. intranasal
Answers: 2


Chemistry, 22.06.2019 12:30, pup88
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Biology, 25.04.2021 17:20

Chemistry, 25.04.2021 17:20

Mathematics, 25.04.2021 17:20


Business, 25.04.2021 17:20


Mathematics, 25.04.2021 17:20


Social Studies, 25.04.2021 17:20






