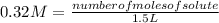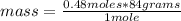Chemistry, 21.10.2020 14:01, moningersavannah
1. Calculate the number of grams of solute required for the preparation of 1.5L of 0.32M NaHCO3 (MW=84) V1 X C1 = V2 X C2

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, alexandroperez13
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 22.06.2019 10:50, lejeanjamespete1
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 23.06.2019 01:00, shartiarahoward
Atoms contain subatomic particles called protons and neutrons. when these protons and neutrons spilt, a lot of energy is released
Answers: 3

Chemistry, 23.06.2019 06:00, mirzakasumovic8926
Which factor is likely to impact the possible number of compounds? presence of unlimited number of elements in the periodic table the inability of atoms to align perfectly with other atoms the ability of all elements to react with every other element all elements being equally reactive
Answers: 2
Do you know the correct answer?
1. Calculate the number of grams of solute required for the preparation of 1.5L of 0.32M NaHCO3 (MW=...
Questions in other subjects:






English, 12.10.2019 05:30

History, 12.10.2019 05:30

English, 12.10.2019 05:30

History, 12.10.2019 05:30


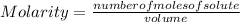
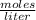 .
.