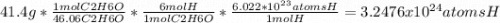Chemistry, 21.10.2020 09:01, joeljuarez128oyusun
Calculate the number of hydrogen atoms in 41.4 g of
ethyl alcohol, C2H60.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, bionicboy03120440
What is the mass of sodium in 3 moles of sodium chloride
Answers: 1

Chemistry, 22.06.2019 06:40, alyons60
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 10:30, jahmira96
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

Chemistry, 23.06.2019 02:30, puppylover72
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2
Do you know the correct answer?
Calculate the number of hydrogen atoms in 41.4 g of
ethyl alcohol, C2H60....
ethyl alcohol, C2H60....
Questions in other subjects:

Mathematics, 13.10.2019 01:30


German, 13.10.2019 01:30

English, 13.10.2019 01:30


English, 13.10.2019 01:30



Mathematics, 13.10.2019 01:30

Mathematics, 13.10.2019 01:30