
Chemistry, 20.10.2020 16:01, Flameking1223
Aspirin is a weak organic acid whose molecular formula is HC9H7O4. An aqueous solution of aspirin is prepared by dissolving 3.60 g/L. The pH of this solution is found to be 2.6. Calculate Ka for aspirin. (atomic mass: C

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, bryneosburn
What’s a special glass that most beakers are made of
Answers: 1

Chemistry, 22.06.2019 13:00, nauticatyson9
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 13:50, awesomegamergurl13
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

Chemistry, 22.06.2019 16:30, sbush1412
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u. s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2
Do you know the correct answer?
Aspirin is a weak organic acid whose molecular formula is HC9H7O4. An aqueous solution of aspirin is...
Questions in other subjects:

English, 25.01.2020 19:31


Mathematics, 25.01.2020 19:31

English, 25.01.2020 19:31


English, 25.01.2020 19:31




English, 25.01.2020 19:31


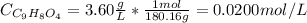
![Ka = \frac{[C_{9}H_{7}O_{4}^{-}][H_{3}O^{+}]}{[C_{9}H_{8}O_{4}]}](/tpl/images/0824/0933/a1a8e.png)
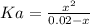
![pH = -log[H_{3}O^{+}]](/tpl/images/0824/0933/b7638.png)
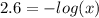
![x = 2.51 \cdot 10^{-3} M = [H_{3}O^{+}] = [C_{9}H_{7}O_{4}^{-}]](/tpl/images/0824/0933/98904.png)
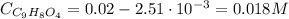
![Ka = \frac{[C_{9}H_{7}O_{4}^{-}][H_{3}O^{+}]}{[C_{9}H_{8}O_{4}]} = \frac{(2.51 \cdot 10^{-3})^{2}}{0.018} = 3.50 \cdot 10^{-4}](/tpl/images/0824/0933/393ec.png)




