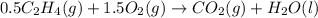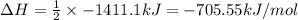Chemistry, 19.10.2020 14:01, samyajones68
Using the reaction below: 2 CO2(g) + 2 H2O(l) → C2H4(g) + 3 O2(g) ΔHrxn= +1411.1 kJ What would be the heat of reaction for this reaction? 0.5 C2H4(g) + 1.5 O2(g) → CO2(g) + H2O(l) ΔHrxn= ??? KJ Question 6 options: a) Not enough information is given b) -2822.2 kJ c) +1411.1 kJ d) -705.55 kJ e) -1411.1 kJ

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, isabelvaldez123
The boiling point of liquids is very high what does it indicate
Answers: 1


Chemistry, 22.06.2019 09:30, strevino9178
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 12:40, valenzueladomipay09u
How does concentration affect reaction rate
Answers: 2
Do you know the correct answer?
Using the reaction below: 2 CO2(g) + 2 H2O(l) → C2H4(g) + 3 O2(g) ΔHrxn= +1411.1 kJ What would be th...
Questions in other subjects:

Mathematics, 04.02.2020 19:49

Mathematics, 04.02.2020 19:49


English, 04.02.2020 19:49


Business, 04.02.2020 19:49

History, 04.02.2020 19:49


Computers and Technology, 04.02.2020 19:49


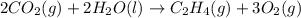
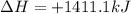
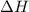
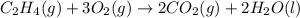
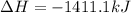
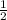 , enthalpy gets half:
, enthalpy gets half: