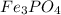Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:30, ajsoccer1705
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3


Chemistry, 22.06.2019 08:30, ebigham5117
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2
Do you know the correct answer?
A 13.58 g sample of a compound contains 8.67 g of iron, Fe, 1.60 g of phosphorus, P, and oxygen, O....
Questions in other subjects:

Social Studies, 03.05.2021 23:50





Biology, 03.05.2021 23:50

Mathematics, 03.05.2021 23:50

Chemistry, 03.05.2021 23:50

Mathematics, 03.05.2021 23:50