
Chemistry, 18.10.2020 14:01, lolagrace06
How many moles of precipitate will be formed when 100.0 mL of 0.200 M NaBr is reacted with excess Pb(NO₃)₂ in the following chemical reaction? 2 NaBr (aq) + Pb(NO₃)₂ (aq) → PbBr₂ (s) + 2 NaNO₃ (aq)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, Cythina2007
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 23.06.2019 02:00, bagofmud8339
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1

Do you know the correct answer?
How many moles of precipitate will be formed when 100.0 mL of 0.200 M NaBr is reacted with excess Pb...
Questions in other subjects:


Mathematics, 20.05.2021 16:40

Mathematics, 20.05.2021 16:40



Mathematics, 20.05.2021 16:40





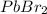 formed will be 0.01 moles.
formed will be 0.01 moles.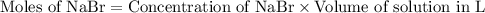
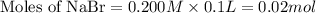
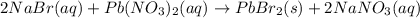
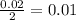 mole of
mole of 



