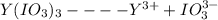Chemistry, 16.10.2020 15:01, kaylamcph7844
The Ksp of yttrium iodate, Y(IO3)3 , is 1.12×10−10 . Calculate the molar solubility, s , of this compound.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, EMQPWE
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1

Chemistry, 22.06.2019 09:30, lisbet123085
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 09:30, psychocatgirl1
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone, due to its high light availability and warm water temperature
Answers: 3
Do you know the correct answer?
The Ksp of yttrium iodate, Y(IO3)3 , is 1.12×10−10 . Calculate the molar solubility, s , of this com...
Questions in other subjects:



Mathematics, 20.11.2020 21:50



Arts, 20.11.2020 21:50


Biology, 20.11.2020 21:50