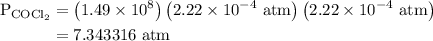Chemistry, 29.09.2019 09:00, charae0185
The kp for the reaction below is 1.49 × 108 at 100.0°c: co(g) + cl2(g) → cocl2(g) in an equilibrium mixture of the three gases, pco = pcl2 = 2.22 × 10-4 atm. the partial pressure of the product, phosgene (cocl2), is atm.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:40, natannale
Darla claims that the first periodic table developed by mendeleev was not completely accurate, so it is not useful at all. harmony argues that it establish the periodic table we use today, making it more credible. who is correct and why? darla is correct, because a model that has any mistakes should be thrown out. darla is correct, because a good model would not need to change. harmony is correct, because mendeleev’s model had all of the information correct in the first version. harmony is correct, because mendeleev’s model made predictions that came true.
Answers: 1

Chemistry, 22.06.2019 11:30, ashleybarrera2000
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

Do you know the correct answer?
The kp for the reaction below is 1.49 × 108 at 100.0°c: co(g) + cl2(g) → cocl2(g) in an equilibrium...
Questions in other subjects:

Arts, 04.12.2020 06:50

Chemistry, 04.12.2020 06:50


Chemistry, 04.12.2020 06:50




English, 04.12.2020 06:50


Mathematics, 04.12.2020 06:50


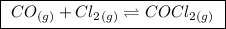 (a balanced reaction)
(a balanced reaction)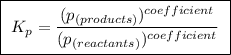
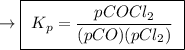
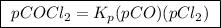
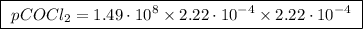
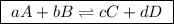 ,
,![\boxed{ \ K_c = \frac{[C]^c[D^d]}{[A]^a[B]^b} \ }](/tpl/images/0273/3667/d057b.png)
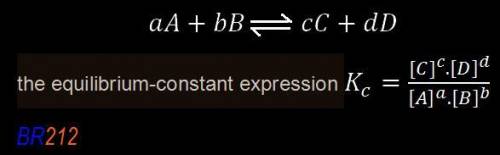
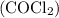 is
is 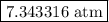 .
.
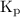 .
.
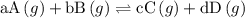
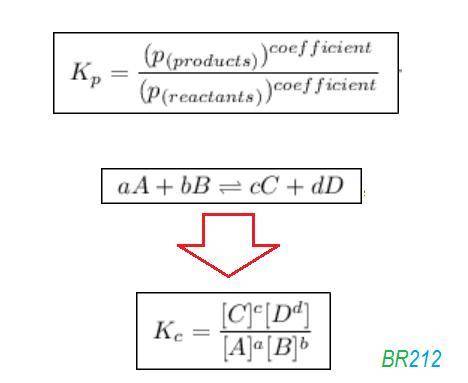
![{{\text{K}}_{\text{p}}} = \dfrac{{{{\left[ {{{\text{P}}_{\text{C}}}} \right]}^{\text{c}}}{{\left[ {{{\text{P}}_{\text{D}}}} \right]}^{\text{d}}}}}{{{{\left[ {{{\text{P}}_{\text{A}}}} \right]}^{\text{a}}}{{\left[ {{{\text{P}}_{\text{B}}}} \right]}^{\text{b}}}}}](/tpl/images/0273/3667/ee3bd.png)
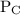 and
and 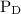 are partial pressures of C and D respectively.
are partial pressures of C and D respectively.
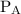 and
and 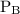 are partial pressures of A and B respectively.
are partial pressures of A and B respectively.
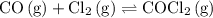
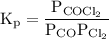 ...... (1)
...... (1)
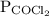 is partial pressure of
is partial pressure of 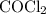 .
.
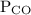 is partial pressure of CO.
is partial pressure of CO.
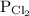 is partial pressure of
is partial pressure of 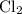 .
.
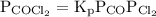 …… (2)
…… (2)
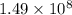 for
for 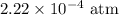 for
for