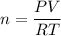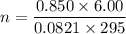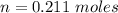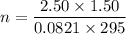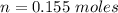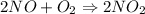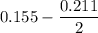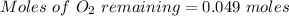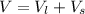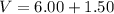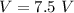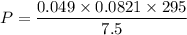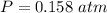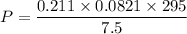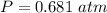Two bulbs are connected by a stopcock. The large bulb, with a volume of 6.00 L, contains nitric oxide at a pressure of 0.850 atm, and the small bulb, with a volume of 1.50 L, contains oxygen at a pressure of 2.50 atm. The temperature at the beginning and the end of the experiment is 22∘C .
After the stopcock is opened, the gases mix and react.
2NO(g)+O2(g)→2NO2(g)
1. Which gases are present at the end of the experiment?
2. What are the partial pressures of the gases? If the gas was consumed completely, put 0 for the answer.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, waterborn7152
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2

Chemistry, 22.06.2019 10:30, tjjjjjjjjjjjjjjjjjjj
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1
Do you know the correct answer?
Two bulbs are connected by a stopcock. The large bulb, with a volume of 6.00 L, contains nitric oxid...
Questions in other subjects: