
Chemistry, 14.10.2020 14:01, adriannacomrosenbark
The rate of reaction in terms of the "rate law expression" includes the rate constant (kk), the concentration of the reactants, and the orders of the reaction with respect to the different reactants. Consider the following reaction: A+B→C+DA+B→C+D The initial concentrations of the reactants AA and BB are 0.200 MM and 0.350 MM, respectively. The rate of reaction is 0.060 M⋅s−1M⋅s−1, and the orders of the reaction, with respect to reactants AA and BB, are 1 and 2, respectively. Determine the rate constant (kk) for the reaction using the rate law. Express your answer in M−2⋅s−1M−2⋅s−1 to three significant figures.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, mosthatedpicky1
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 14:50, rebeccamckellpidge
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 23.06.2019 01:00, shartiarahoward
Atoms contain subatomic particles called protons and neutrons. when these protons and neutrons spilt, a lot of energy is released
Answers: 3
Do you know the correct answer?
The rate of reaction in terms of the "rate law expression" includes the rate constant (kk), the conc...
Questions in other subjects:

Mathematics, 08.06.2021 02:20


Mathematics, 08.06.2021 02:20

Spanish, 08.06.2021 02:20


Business, 08.06.2021 02:20


Mathematics, 08.06.2021 02:20

History, 08.06.2021 02:20


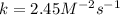
![r=k[A][B]^2](/tpl/images/0805/6865/487f0.png)
![k=\frac{r}{[A][B]^2}\\ \\k=\frac{0.060M/s}{(0.200M)(0.350M)^2} \\\\k=2.45M^{-2}s^{-1}](/tpl/images/0805/6865/72630.png)




