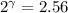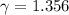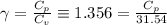The constant volume heat capacity of a gas can be measured by observing the decrease in temperature when it expands adiabatically and reversibly. If the decrease in pressure is also measured, we can use it to infer the value of γ = Cp/Cv and hence, by combining the two values, deduce the constant-pressure heat capacity. A fluorocarbon gas was allowed to expand reversibly and adiabatically to twice its volume; as a result, the temperature fell from 298.15 K to 248.44 K and its pressure fell from 202.94 kPa to 81.840 kPa. Evaluate Cp

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 20:10, sarahalexa19
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 23.06.2019 00:50, alainacorkell6472
What is the enthalpy of combustion (per mole) of c4h10 (g)? –2,657.5 kj/mol –5315.0 kj/mol –509.7 kj/mol –254.8 kj/mol
Answers: 1

Chemistry, 23.06.2019 01:30, kenldykido2300
Adirect relationship can be represented by: a curve a pie chart
Answers: 2
Do you know the correct answer?
The constant volume heat capacity of a gas can be measured by observing the decrease in temperature...
Questions in other subjects:


Biology, 20.05.2021 18:30

Mathematics, 20.05.2021 18:30

Social Studies, 20.05.2021 18:30

Mathematics, 20.05.2021 18:30

Mathematics, 20.05.2021 18:30


Mathematics, 20.05.2021 18:30

Mathematics, 20.05.2021 18:30


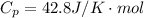
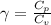
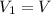

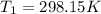
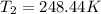
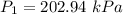
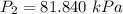
![T_2 = T_1 * [ \frac{V_1}{V_2} ]^{\frac{R}{C_v} }](/tpl/images/0802/4357/d7f78.png)
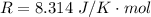
![248.44 = 298.15 * [ \frac{V}{2V} ]^{\frac{8.314}{C_v} }](/tpl/images/0802/4357/a72d7.png)
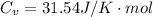
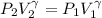
![[ 81.840 *10^3] [2V]^{\gamma} = [202.94 *10^3] V^{\gamma}](/tpl/images/0802/4357/43b7e.png)