
Chemistry, 11.10.2020 01:01, officialrogerfp3gf2s
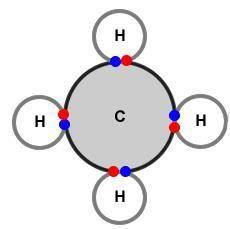
What conclusion about bonding can be drawn from these diagrams?
A) In ionic bonding the valence electrons are combined while the valence electrons are electrostatically portioned during non-polar covalent bonding.
B) The valence electrons are shared between both atoms in covalent bonding while the valence electrons are completely transferred in ionic bonding.
C) Valence electrons reside in a sea of electrons in metallic bonding while valence electrons are permanently exchanged during crystalline bonding.
D) During amorphous bonding the valence electrons are unevenly shared between the atoms while the valence electrons are borrowed in polar covalent bonding.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, caeyanij
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3


Chemistry, 22.06.2019 14:30, lorrainelopez
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1
Do you know the correct answer?
What conclusion about bonding can be drawn from these diagrams?
A) In ionic bonding the valence ele...
Questions in other subjects:

Mathematics, 10.12.2020 05:30

English, 10.12.2020 05:30





Mathematics, 10.12.2020 05:30

English, 10.12.2020 05:30

History, 10.12.2020 05:30

Mathematics, 10.12.2020 05:30






