Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, nikejose11
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3

Chemistry, 23.06.2019 04:31, cassiuspricerules
What is the amount of energy for a photon that has a 125 cm wavelength
Answers: 2

Chemistry, 23.06.2019 05:00, rosezgomez97
Asolution is made by dissolving 2.3 moles of sodium chloride (nacl) in 0.155 kilograms of water. if the molal boiling point constant for water (kb) is 0.51 °c/m, what would be the boiling point of this solution? show all the steps taken to solve this problem.
Answers: 1

Chemistry, 23.06.2019 13:30, bossboybaker
What happens to acetone molecules when you add heat to a beaker of liquid acetone?
Answers: 1
Do you know the correct answer?
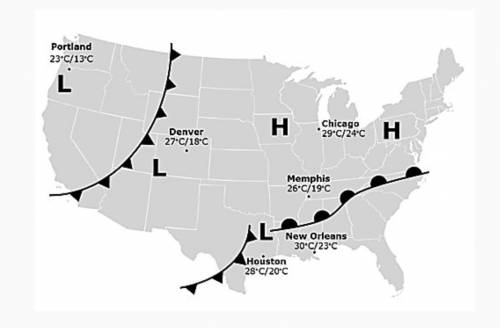
If the weather map above is from today’s forecast, which city is most likely to see its temperatures...
Questions in other subjects:



Chemistry, 18.03.2021 02:20





Computers and Technology, 18.03.2021 02:20

Mathematics, 18.03.2021 02:20