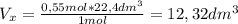Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:50, AysiaRamosLee
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 09:00, triddi666
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 22.06.2019 11:10, hannah2757
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1
Do you know the correct answer?
Use the equation below to determine what volume of nitrous oxide (n20) can be produced from the deco...
Questions in other subjects:






Geography, 16.10.2020 07:01

Mathematics, 16.10.2020 07:01


Mathematics, 16.10.2020 07:01