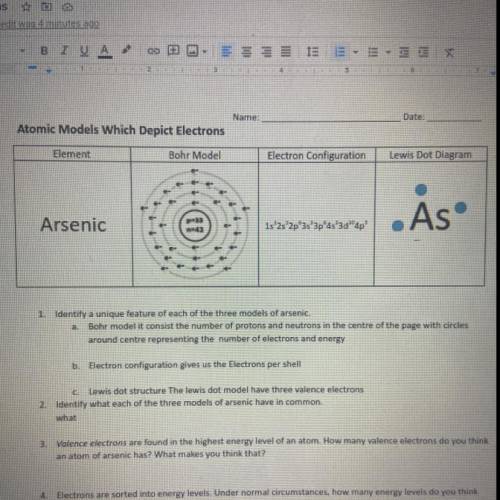
Atomic Models Which Depict Electrons
Element
Bohr Model
Electron Configuration
Le...

Chemistry, 29.09.2020 20:01, queenkendra16
Atomic Models Which Depict Electrons
Element
Bohr Model
Electron Configuration
Lewis Dot Diagram
. As
'
Arsenic
1s 2s 2p 3s 3pas'3d"
ap'
1. Identify a unique feature of each of the three models of arsenie.
Bohr modelit consist the number of protons and neutrons in the centre of the page with circles
around contre representing number of electrons and energy
a
D: Electron configuration gives us the Electrons per shell
Lewis dot structure The lewis dot model have three valence electrons
2. Identify what each of the three models of arsenic have in common
what
Valence electrons are found in the highest energy level of an atom. How many valence electrons do you think
an atom of arsenic has? What makes you think that?
4. Electrons are sorted into energy levels. Under normal circumstances, how many energy levels do you think
contain electrons in an atom of arsenic? What makes you think that?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:30, mastershadow2018
Agroup of students is studying convection currents. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other in an area with warm air. after 10 minutes, the balloons are released from a height of 1 meter. which of the following do the students most likely observe? a. the balloons both rise. the cold balloon is larger than the warm balloon. b. the balloons rise at the same rate. both balloons are the same size. c. the warm balloon expands and rises. the cold balloon shrinks and sinks. d. the cold balloon expands and rises. the warm balloon shrinks and sinks.
Answers: 2

Chemistry, 22.06.2019 12:30, UaRemomGAY
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Do you know the correct answer?
Questions in other subjects:















