Can anyone please help with this
...

Answers: 1
Other questions on the subject: Chemistry



Do you know the correct answer?
Questions in other subjects:

Mathematics, 24.03.2021 21:00




Mathematics, 24.03.2021 21:00

Mathematics, 24.03.2021 21:00




Computers and Technology, 24.03.2021 21:00



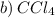 is the correct choice.
is the correct choice.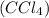 is a non-polar solvent. Therefore, the non-polar substance will be most soluble in
is a non-polar solvent. Therefore, the non-polar substance will be most soluble in 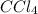 .
.



