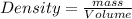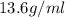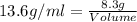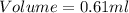Chemistry, 26.08.2019 09:00, connersitte1221l
Athermometer containing 8.3g of mercury has broken. if mercury ha a density of 13.6g/ml. what volume is spilled?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:50, lejeanjamespete1
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1


Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
Do you know the correct answer?
Athermometer containing 8.3g of mercury has broken. if mercury ha a density of 13.6g/ml. what volume...
Questions in other subjects:



Mathematics, 24.02.2021 18:50

Mathematics, 24.02.2021 18:50

Spanish, 24.02.2021 18:50


Arts, 24.02.2021 18:50

Mathematics, 24.02.2021 18:50

Mathematics, 24.02.2021 18:50

Law, 24.02.2021 18:50