
Chemistry, 23.09.2020 14:01, tiwaribianca475
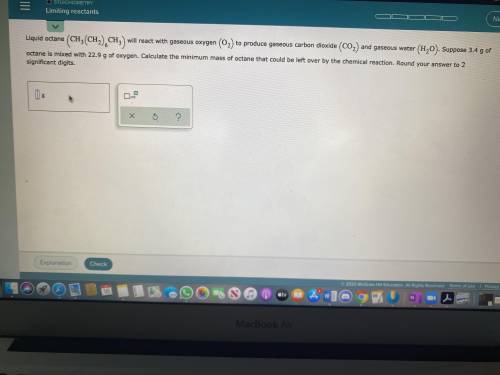
Liquid octane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water. Suppose 3.4g of octane is mixed with 22.9g of oxygen. Calculate the minimum mass of octane that could be left over by the chemical reaction. Round your answer to 2 significant digits.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, thatonestudent2271
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 05:30, alexusnicole817
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 23.06.2019 00:30, kylee65
What are the advantages of using the metric system? designed as a decimal system making conversions simpler more accurate system of measurement has prefixes that correspond to an amount to use with all base units used by the entire scientific community
Answers: 2

Chemistry, 23.06.2019 01:00, daniel1480
Which fossil fuel is mainly used for heating and cooking? a. electricity b. coal c. petroleum d. natural gas
Answers: 2
Do you know the correct answer?
Liquid octane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water. Su...
Questions in other subjects:

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Mathematics, 13.09.2020 05:01

Chemistry, 13.09.2020 05:01






